
 |
Our Research"Worms smell and taste just like humans" We are using a worm, the tiny soil nematode Caenorhabditis elegans,
as a model system to study nervous system development per se and also
as it relates to humans. FOR THE RECORD: Peter Swoboda does not like the buttery smell of cinema
pop-corn (di-acetyl). |
"The development of sensory structures in C. elegans" Cilia and flagella are evolutionarily conserved subcellular organelles functioning in cell motility, movement of extracellular fluids, sensory perception (e.g. smell and vision) and the determination of left-right asymmetry. They occur on many, many different cell types across a large variety of eukaryotic organisms. While a great deal is known about the structure, function and motility of cilia, very little is known about the molecular mechanisms that regulate ciliogenesis in a cell-type specific and developmental manner. How do cilia become functional? How can they do what they do? One way to address these questions is by using sensory cilium formation in the nematode Caenorhabditis elegans as a model system. Sixty neurons of the hermaphrodite worm form various structurally distinct types of sensory endings. Sensory cilia are the only types of cilia in the worm, making C. elegans a homogeneous experimental system. Grounded in an extraordinary database (e.g. complete genome sequence, cell lineage, and nervous system structure are known), the C. elegans model system is readily suited for a multi-faceted approach that is based on mutational analyses of genes involved in sensory cilium formation. Genetic screens for altered sensory behavior and altered sensory neuron morphology undertaken in many laboratories have identified a large group of mutants with morphological defects in their sensory cilia. A variety of genetic, genomic, molecular, cell biological, biochemical and ultrastructural methods are available to allow detailed characterization of mutant phenotypes. Because the structural and functional characteristics of C. elegans sensory cilia are very similar to those in mammals, results obtained with the model system C. elegans will have general significance. |
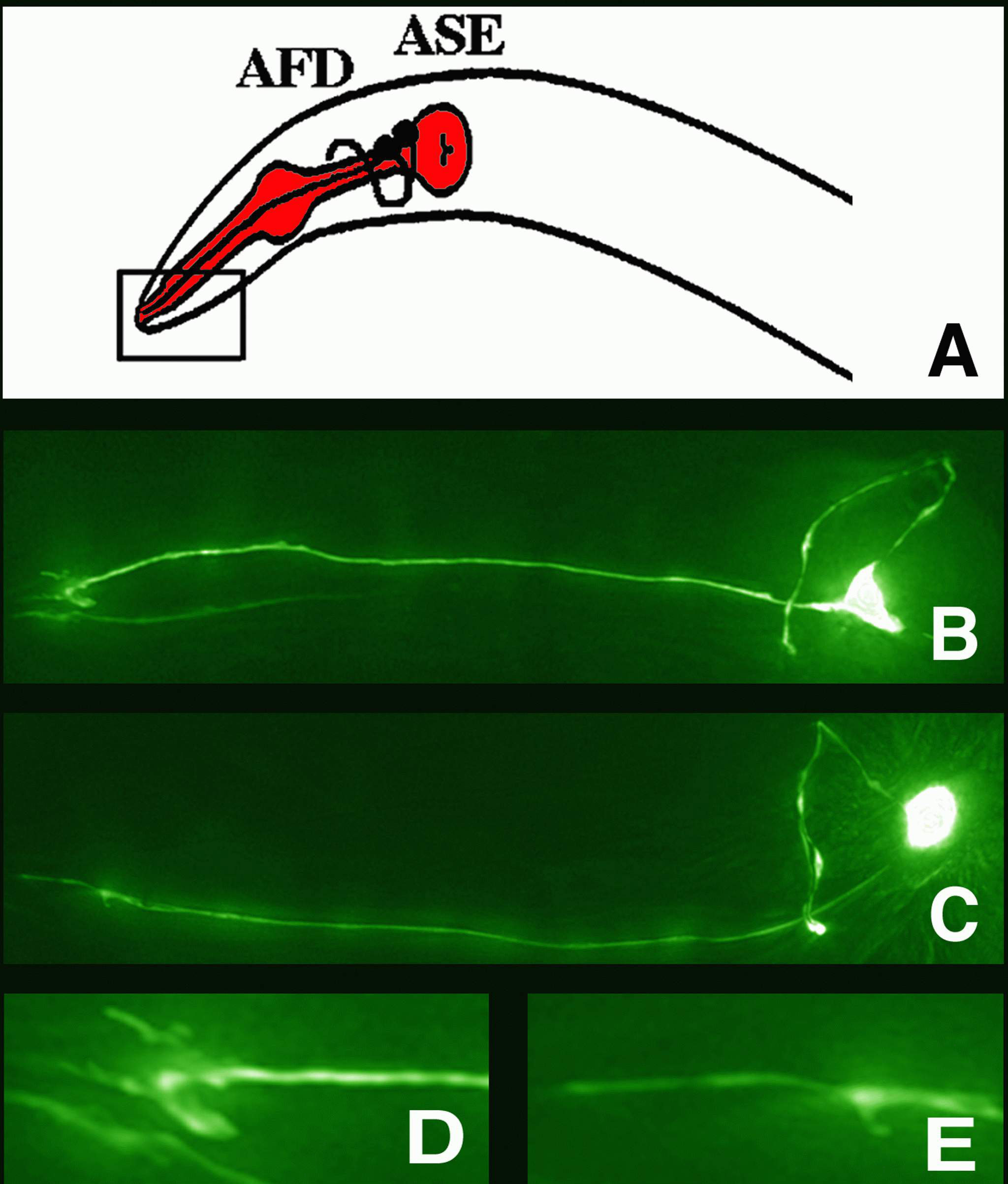
Figure 1: Worm sensory neurons
(A) A schematic showing the head of a worm with its pharynx (mouth) colored in red.
Two sensory neurons (AFD and ASE) are shown as black dots with their sensory processes
(dendrites; straight lines) extending toward the tip of the nose (rectangle), and their other
processes extending into the "brain" of the worm (axons; S-shaped lines between the bulbs
of the pharynx). (B) The neuron AFD that is used for temperature sensation.
Its specialized sensory ending (fan-like microvilli) at the left is also shown
enlarged in (D). (C) The neuron ASE that is used for chemosensation (taste).
Its specialized sensory ending (rod-like cilium) at the left is also shown enlarged
in (E). | |
We have previously shown that the gene daf-19 is essential for sensory neuron ciliogenesis in C. elegans. It encodes a member of the RFX family of transcription factors. By acting through its target site in promoter regions, the x-box, DAF-19 appears to be the main regulator of a number of effector genes involved in sensory cilium formation and function (che-2, che-13, odr-4, osm-1, osm-5, osm-6, xbx-1). One of our major aims is to define the complete set of DAF-19 target genes coding for cilia components and to determine their structural/functional roles in cilia formation. This is based on ongoing genome-wide screens in C. elegans, C. briggsae, Drosophila and other organisms for genes containing consensus DAF-19 or RFX-type binding sites in their regulatory regions. Candidate genes (xbx genes) are being validated in C. elegans by examining their expression in daf-19 mutants. Their role in cilia is being addressed by using RNA interference, by generating deletion mutants (with the help of the C. elegans Gene Knock-Out Facility) and by interaction screens using already known cilia components. Additional aims are a detailed structure-function analysis of DAF-19 and the identification of factors that cooperate with DAF-19 in ciliated sensory neuron differentiation with the help of genetic screens. As daf-19 likely acts very early in the hierarchy of events inducing ciliogenesis, it might be regarded as a determination gene for cilium formation. In vivo misexpression studies of DAF-19 have been initiated. The introduction of DAF-19 into usually non-ciliated cells as well as de-regulated expression in sensory neurons will provide more information about the potential of this transcription factor as a determination gene for cilium formation. Furthermore we try to determine the time window during which a developing sensory neuron can become a ciliated neuron. Since cilia are cellular compartments devoid of protein synthesis, we are also working on elucidating the machinery that exists to transport components to, from and within cilia (intraflagellar transport - IFT). | |
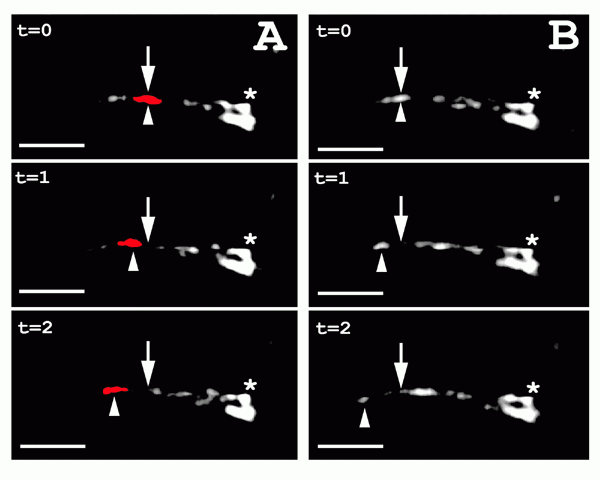
Figure 2: Intraflagellar transport in worm sensory cilia.
Movement of OSM-5::GFP in sensory cilia. Two sequences (A and B) from the same
worm are shown, each composed of three sequential frames. The tip of
the cilium is toward the left, while the transition zone that links
the cilium to the dendritic process of the neuron is shown at the
right (asterisk). The arrow indicates the initial position of the
fluorescent particle at time 0 in all frames. The arrowhead indicates
the location of the fluorescent particle in each frame. For additional
clarity one of the moving fluorescent particles is colored in red in
the panels on the left. | |
To further characterize the set of genes that comprise the
cilium formation module we are (in collaboration with computer scientists)
searching through promoter sequences of selected subsets of cilium specific
and xbx genes for other conserved promoter elements using C. elegans and
various other (including human) genome information.
Elucidation of the conserved regulatory pathways controlling ciliogenesis will advance our basic knowledge on a highly conserved organelle ("after all, worms smell and taste just like humans") and will contribute to our understanding of human pathologies implicating cilia: e.g. primary ciliary dyskinesia (PCD), polycystic kidney disease (PKD), retinal degradation syndromes or the Bardet-Biedl syndrome (BBS). |